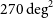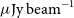36 results
Development, implementation, and dissemination of operational innovations across the trial innovation network
- Marisha E. Palm, Terri L. Edwards, Cortney Wieber, Marie T. Kay, Eve Marion, Leslie Boone, Angeline Nanni, Michelle Jones, Eilene Pham, Meghan Hildreth, Karen Lane, Nichol McBee, Daniel K. Benjamin, Jr, Gordon R. Bernard, J. Michael Dean, Jamie P. Dwyer, Daniel E. Ford, Daniel F. Hanley, Paul A. Harris, Consuelo H. Wilkins, Harry P. Selker
-
- Journal:
- Journal of Clinical and Translational Science / Volume 7 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 20 October 2023, e251
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Improving the quality and conduct of multi-center clinical trials is essential to the generation of generalizable knowledge about the safety and efficacy of healthcare treatments. Despite significant effort and expense, many clinical trials are unsuccessful. The National Center for Advancing Translational Science launched the Trial Innovation Network to address critical roadblocks in multi-center trials by leveraging existing infrastructure and developing operational innovations. We provide an overview of the roadblocks that led to opportunities for operational innovation, our work to develop, define, and map innovations across the network, and how we implemented and disseminated mature innovations.
Decentralized clinical trials in the trial innovation network: Value, strategies, and lessons learned
- Daniel F. Hanley, Jr, Gordon R. Bernard, Consuelo H. Wilkins, Harry P. Selker, Jamie P. Dwyer, J. Michael Dean, Daniel Kelly Benjamin, Jr, Sarah E. Dunsmore, Salina P. Waddy, Kenneth L. Wiley, Jr, Marisha E. Palm, W. Andrew Mould, Daniel F. Ford, Jeri S. Burr, Jacqueline Huvane, Karen Lane, Lori Poole, Terri L. Edwards, Nan Kennedy, Leslie R. Boone, Jasmine Bell, Emily Serdoz, Loretta M. Byrne, Paul A. Harris
-
- Journal:
- Journal of Clinical and Translational Science / Volume 7 / Issue 1 / 2023
- Published online by Cambridge University Press:
- 25 July 2023, e170
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
New technologies and disruptions related to Coronavirus disease-2019 have led to expansion of decentralized approaches to clinical trials. Remote tools and methods hold promise for increasing trial efficiency and reducing burdens and barriers by facilitating participation outside of traditional clinical settings and taking studies directly to participants. The Trial Innovation Network, established in 2016 by the National Center for Advancing Clinical and Translational Science to address critical roadblocks in clinical research and accelerate the translational research process, has consulted on over 400 research study proposals to date. Its recommendations for decentralized approaches have included eConsent, participant-informed study design, remote intervention, study task reminders, social media recruitment, and return of results for participants. Some clinical trial elements have worked well when decentralized, while others, including remote recruitment and patient monitoring, need further refinement and assessment to determine their value. Partially decentralized, or “hybrid” trials, offer a first step to optimizing remote methods. Decentralized processes demonstrate potential to improve urban-rural diversity, but their impact on inclusion of racially and ethnically marginalized populations requires further study. To optimize inclusive participation in decentralized clinical trials, efforts must be made to build trust among marginalized communities, and to ensure access to remote technology.
The Evolutionary Map of the Universe Pilot Survey – ADDENDUM
- Ray P. Norris, Joshua Marvil, J. D. Collier, Anna D. Kapińska, Andrew N. O’Brien, L. Rudnick, Heinz Andernach, Jacobo Asorey, Michael J. I. Brown, Marcus Brüggen, Evan Crawford, Jayanne English, Syed Faisal ur Rahman, Miroslav D. Filipović, Yjan Gordon, Gülay Gürkan, Catherine Hale, Andrew M. Hopkins, Minh T. Huynh, Kim HyeongHan, M. James Jee, Bärbel S. Koribalski, Emil Lenc, Kieran Luken, David Parkinson, Isabella Prandoni, Wasim Raja, Thomas H. Reiprich, Christopher J. Riseley, Stanislav S. Shabala, Jaimie R. Sheil, Tessa Vernstrom, Matthew T. Whiting, James R. Allison, C. S. Anderson, Lewis Ball, Martin Bell, John Bunton, T. J. Galvin, Neeraj Gupta, Aidan Hotan, Colin Jacka, Peter J. Macgregor, Elizabeth K. Mahony, Umberto Maio, Vanessa Moss, M. Pandey-Pommier, Maxim A. Voronkov
-
- Journal:
- Publications of the Astronomical Society of Australia / Volume 39 / 2022
- Published online by Cambridge University Press:
- 02 November 2022, e055
-
- Article
- Export citation
The prescriber’s guide to classic MAO inhibitors (phenelzine, tranylcypromine, isocarboxazid) for treatment-resistant depression
- Vincent Van den Eynde, Wegdan R. Abdelmoemin, Magid M. Abraham, Jay D. Amsterdam, Ian M. Anderson, Chittaranjan Andrade, Glen B. Baker, Aartjan T.F. Beekman, Michael Berk, Tom K. Birkenhäger, Barry B. Blackwell, Pierre Blier, Marc B.J. Blom, Alexander J. Bodkin, Carlo I. Cattaneo, Bezalel Dantz, Jonathan Davidson, Boadie W. Dunlop, Ryan F. Estévez, Shalom S. Feinberg, John P.M. Finberg, Laura J. Fochtmann, David Gotlib, Andrew Holt, Thomas R. Insel, Jens K. Larsen, Rajnish Mago, David B. Menkes, Jonathan M. Meyer, David J. Nutt, Gordon Parker, Mark D. Rego, Elliott Richelson, Henricus G. Ruhé, Jerónimo Sáiz-Ruiz, Stephen M. Stahl, Thomas Steele, Michael E. Thase, Sven Ulrich, Anton J.L.M. van Balkom, Eduard Vieta, Ian Whyte, Allan H. Young, Peter K. Gillman
-
- Journal:
- CNS Spectrums / Volume 28 / Issue 4 / August 2023
- Published online by Cambridge University Press:
- 15 July 2022, pp. 427-440
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
This article is a clinical guide which discusses the “state-of-the-art” usage of the classic monoamine oxidase inhibitor (MAOI) antidepressants (phenelzine, tranylcypromine, and isocarboxazid) in modern psychiatric practice. The guide is for all clinicians, including those who may not be experienced MAOI prescribers. It discusses indications, drug-drug interactions, side-effect management, and the safety of various augmentation strategies. There is a clear and broad consensus (more than 70 international expert endorsers), based on 6 decades of experience, for the recommendations herein exposited. They are based on empirical evidence and expert opinion—this guide is presented as a new specialist-consensus standard. The guide provides practical clinical advice, and is the basis for the rational use of these drugs, particularly because it improves and updates knowledge, and corrects the various misconceptions that have hitherto been prominent in the literature, partly due to insufficient knowledge of pharmacology. The guide suggests that MAOIs should always be considered in cases of treatment-resistant depression (including those melancholic in nature), and prior to electroconvulsive therapy—while taking into account of patient preference. In selected cases, they may be considered earlier in the treatment algorithm than has previously been customary, and should not be regarded as drugs of last resort; they may prove decisively effective when many other treatments have failed. The guide clarifies key points on the concomitant use of incorrectly proscribed drugs such as methylphenidate and some tricyclic antidepressants. It also illustrates the straightforward “bridging” methods that may be used to transition simply and safely from other antidepressants to MAOIs.
276 Bilirubin Oxidative Products as Predictive Biomarkers of Cerebral Vasospasm: A Pilot Study
- Stefano H Byer, Shweta Goswami, Wes Gordon, Michael G Abraham
-
- Journal:
- Journal of Clinical and Translational Science / Volume 6 / Issue s1 / April 2022
- Published online by Cambridge University Press:
- 19 April 2022, p. 46
-
- Article
-
- You have access Access
- Open access
- Export citation
-
OBJECTIVES/GOALS: Our aim is to understand the role bilirubin oxidation products play in the development of cerebral vasospasm in patients with subarachnoid hemorrhage. We aim to evaluate the time course of bilirubin, HO-1, and SOD1 in relation to the subsequent development of vasospasm in to establish predictors of vasospasm development. METHODS/STUDY POPULATION: Prospective cohort observational study involving collection of CSF samples of pts admitted to KU NeuroICU with SAH and placement of EVD. CSF will be extracted from the EVD of patients on the day of placement of the EVD, and then each subsequent day for a total of 10 days. Bilirubin concentration will be determined by means of spectrophotometry. HO-1 will be measured using a commercially available ELISA kit. Cu/Zn-Superoxide Dismutase will be measured using a commercially available ELISA kit. A review of patients chart will then be performed following discharge from hospital to determine if a diagnosis of vasospasm was made, details of the vasospasm (i.e. symptoms, severity), as well as to obtain demographic data and events occurring during patients admission that could confound statistical analysis. RESULTS/ANTICIPATED RESULTS: First: we will investigate the feasibility of collecting serial CSF samples and processing them for target analyte quantification. We predict that the protocol will yield quality data that will result in insight on the pathophysiology of cerebral vasospasm. Second: we will characterize the changes in target CSF bilirubin breakdown analytes over 10 days. From this we hypothesize that as bilirubin oxidation increases, the propensity for cerebral vasospasm will also increase. DISCUSSION/SIGNIFICANCE: If there is a clear correlation between formation of bilirubin and increase in HO-1, and SOD1, with the clinical signs of vasospasm, this could be used as a biomarker for not only identifying patients at risk for developing these complications but also a means to follow the effectiveness of potential therapies.
The Evolutionary Map of the Universe pilot survey
- Part of
- Ray P. Norris, Joshua Marvil, J. D. Collier, Anna D. Kapińska, Andrew N. O’Brien, L. Rudnick, Heinz Andernach, Jacobo Asorey, Michael J. I. Brown, Marcus Brüggen, Evan Crawford, Jayanne English, Syed Faisal ur Rahman, Miroslav D. Filipović, Yjan Gordon, Gülay Gürkan, Catherine Hale, Andrew M. Hopkins, Minh T. Huynh, Kim HyeongHan, M. James Jee, Bärbel S. Koribalski, Emil Lenc, Kieran Luken, David Parkinson, Isabella Prandoni, Wasim Raja, Thomas H. Reiprich, Christopher J. Riseley, Stanislav S. Shabala, Jaimie R. Sheil, Tessa Vernstrom, Matthew T. Whiting, James R. Allison, C. S. Anderson, Lewis Ball, Martin Bell, John Bunton, T. J. Galvin, Neeraj Gupta, Aidan Hotan, Colin Jacka, Peter J. Macgregor, Elizabeth K. Mahony, Umberto Maio, Vanessa Moss, M. Pandey-Pommier, Maxim A. Voronkov
-
- Journal:
- Publications of the Astronomical Society of Australia / Volume 38 / 2021
- Published online by Cambridge University Press:
- 07 September 2021, e046
-
- Article
-
- You have access Access
- HTML
- Export citation
-
We present the data and initial results from the first pilot survey of the Evolutionary Map of the Universe (EMU), observed at 944 MHz with the Australian Square Kilometre Array Pathfinder (ASKAP) telescope. The survey covers
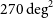 $270 \,\mathrm{deg}^2$
of an area covered by the Dark Energy Survey, reaching a depth of 25–30
$270 \,\mathrm{deg}^2$
of an area covered by the Dark Energy Survey, reaching a depth of 25–30
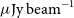 $\mu\mathrm{Jy\ beam}^{-1}$
rms at a spatial resolution of
$\mu\mathrm{Jy\ beam}^{-1}$
rms at a spatial resolution of
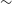 $\sim$
11–18 arcsec, resulting in a catalogue of
$\sim$
11–18 arcsec, resulting in a catalogue of
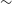 $\sim$
220 000 sources, of which
$\sim$
220 000 sources, of which
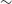 $\sim$
180 000 are single-component sources. Here we present the catalogue of single-component sources, together with (where available) optical and infrared cross-identifications, classifications, and redshifts. This survey explores a new region of parameter space compared to previous surveys. Specifically, the EMU Pilot Survey has a high density of sources, and also a high sensitivity to low surface brightness emission. These properties result in the detection of types of sources that were rarely seen in or absent from previous surveys. We present some of these new results here.
$\sim$
180 000 are single-component sources. Here we present the catalogue of single-component sources, together with (where available) optical and infrared cross-identifications, classifications, and redshifts. This survey explores a new region of parameter space compared to previous surveys. Specifically, the EMU Pilot Survey has a high density of sources, and also a high sensitivity to low surface brightness emission. These properties result in the detection of types of sources that were rarely seen in or absent from previous surveys. We present some of these new results here.
Characterisation of age and polarity at onset in bipolar disorder
- Janos L. Kalman, Loes M. Olde Loohuis, Annabel Vreeker, Andrew McQuillin, Eli A. Stahl, Douglas Ruderfer, Maria Grigoroiu-Serbanescu, Georgia Panagiotaropoulou, Stephan Ripke, Tim B. Bigdeli, Frederike Stein, Tina Meller, Susanne Meinert, Helena Pelin, Fabian Streit, Sergi Papiol, Mark J. Adams, Rolf Adolfsson, Kristina Adorjan, Ingrid Agartz, Sofie R. Aminoff, Heike Anderson-Schmidt, Ole A. Andreassen, Raffaella Ardau, Jean-Michel Aubry, Ceylan Balaban, Nicholas Bass, Bernhard T. Baune, Frank Bellivier, Antoni Benabarre, Susanne Bengesser, Wade H Berrettini, Marco P. Boks, Evelyn J. Bromet, Katharina Brosch, Monika Budde, William Byerley, Pablo Cervantes, Catina Chillotti, Sven Cichon, Scott R. Clark, Ashley L. Comes, Aiden Corvin, William Coryell, Nick Craddock, David W. Craig, Paul E. Croarkin, Cristiana Cruceanu, Piotr M. Czerski, Nina Dalkner, Udo Dannlowski, Franziska Degenhardt, Maria Del Zompo, J. Raymond DePaulo, Srdjan Djurovic, Howard J. Edenberg, Mariam Al Eissa, Torbjørn Elvsåshagen, Bruno Etain, Ayman H. Fanous, Frederike Fellendorf, Alessia Fiorentino, Andreas J. Forstner, Mark A. Frye, Janice M. Fullerton, Katrin Gade, Julie Garnham, Elliot Gershon, Michael Gill, Fernando S. Goes, Katherine Gordon-Smith, Paul Grof, Jose Guzman-Parra, Tim Hahn, Roland Hasler, Maria Heilbronner, Urs Heilbronner, Stephane Jamain, Esther Jimenez, Ian Jones, Lisa Jones, Lina Jonsson, Rene S. Kahn, John R. Kelsoe, James L. Kennedy, Tilo Kircher, George Kirov, Sarah Kittel-Schneider, Farah Klöhn-Saghatolislam, James A. Knowles, Thorsten M. Kranz, Trine Vik Lagerberg, Mikael Landen, William B. Lawson, Marion Leboyer, Qingqin S. Li, Mario Maj, Dolores Malaspina, Mirko Manchia, Fermin Mayoral, Susan L. McElroy, Melvin G. McInnis, Andrew M. McIntosh, Helena Medeiros, Ingrid Melle, Vihra Milanova, Philip B. Mitchell, Palmiero Monteleone, Alessio Maria Monteleone, Markus M. Nöthen, Tomas Novak, John I. Nurnberger, Niamh O'Brien, Kevin S. O'Connell, Claire O'Donovan, Michael C. O'Donovan, Nils Opel, Abigail Ortiz, Michael J. Owen, Erik Pålsson, Carlos Pato, Michele T. Pato, Joanna Pawlak, Julia-Katharina Pfarr, Claudia Pisanu, James B. Potash, Mark H Rapaport, Daniela Reich-Erkelenz, Andreas Reif, Eva Reininghaus, Jonathan Repple, Hélène Richard-Lepouriel, Marcella Rietschel, Kai Ringwald, Gloria Roberts, Guy Rouleau, Sabrina Schaupp, William A Scheftner, Simon Schmitt, Peter R. Schofield, K. Oliver Schubert, Eva C. Schulte, Barbara Schweizer, Fanny Senner, Giovanni Severino, Sally Sharp, Claire Slaney, Olav B. Smeland, Janet L. Sobell, Alessio Squassina, Pavla Stopkova, John Strauss, Alfonso Tortorella, Gustavo Turecki, Joanna Twarowska-Hauser, Marin Veldic, Eduard Vieta, John B. Vincent, Wei Xu, Clement C. Zai, Peter P. Zandi, Psychiatric Genomics Consortium (PGC) Bipolar Disorder Working Group, International Consortium on Lithium Genetics (ConLiGen), Colombia-US Cross Disorder Collaboration in Psychiatric Genetics, Arianna Di Florio, Jordan W. Smoller, Joanna M. Biernacka, Francis J. McMahon, Martin Alda, Bertram Müller-Myhsok, Nikolaos Koutsouleris, Peter Falkai, Nelson B. Freimer, Till F.M. Andlauer, Thomas G. Schulze, Roel A. Ophoff
-
- Journal:
- The British Journal of Psychiatry / Volume 219 / Issue 6 / December 2021
- Published online by Cambridge University Press:
- 25 August 2021, pp. 659-669
- Print publication:
- December 2021
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Background
Studying phenotypic and genetic characteristics of age at onset (AAO) and polarity at onset (PAO) in bipolar disorder can provide new insights into disease pathology and facilitate the development of screening tools.
AimsTo examine the genetic architecture of AAO and PAO and their association with bipolar disorder disease characteristics.
MethodGenome-wide association studies (GWASs) and polygenic score (PGS) analyses of AAO (n = 12 977) and PAO (n = 6773) were conducted in patients with bipolar disorder from 34 cohorts and a replication sample (n = 2237). The association of onset with disease characteristics was investigated in two of these cohorts.
ResultsEarlier AAO was associated with a higher probability of psychotic symptoms, suicidality, lower educational attainment, not living together and fewer episodes. Depressive onset correlated with suicidality and manic onset correlated with delusions and manic episodes. Systematic differences in AAO between cohorts and continents of origin were observed. This was also reflected in single-nucleotide variant-based heritability estimates, with higher heritabilities for stricter onset definitions. Increased PGS for autism spectrum disorder (β = −0.34 years, s.e. = 0.08), major depression (β = −0.34 years, s.e. = 0.08), schizophrenia (β = −0.39 years, s.e. = 0.08), and educational attainment (β = −0.31 years, s.e. = 0.08) were associated with an earlier AAO. The AAO GWAS identified one significant locus, but this finding did not replicate. Neither GWAS nor PGS analyses yielded significant associations with PAO.
ConclusionsAAO and PAO are associated with indicators of bipolar disorder severity. Individuals with an earlier onset show an increased polygenic liability for a broad spectrum of psychiatric traits. Systematic differences in AAO across cohorts, continents and phenotype definitions introduce significant heterogeneity, affecting analyses.
Response of the trial innovation network to the COVID-19 pandemic
- Rachel G. Greenberg, Lori Poole, Daniel E. Ford, Daniel Hanley, Harry P. Selker, Karen Lane, J. Michael Dean, Jeri Burr, Paul Harris, Consuelo H. Wilkins, Gordon Bernard, Terri Edwards, Daniel K. Benjamin, Jr
-
- Journal:
- Journal of Clinical and Translational Science / Volume 5 / Issue 1 / 2021
- Published online by Cambridge University Press:
- 20 April 2021, e100
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Introduction:
The COVID-19 pandemic prompted the development and implementation of hundreds of clinical trials across the USA. The Trial Innovation Network (TIN), funded by the National Center for Advancing Translational Sciences, was an established clinical research network that pivoted to respond to the pandemic.
Methods:The TIN’s three Trial Innovation Centers, Recruitment Innovation Center, and 66 Clinical and Translational Science Award Hub institutions, collaborated to adapt to the pandemic’s rapidly changing landscape, playing central roles in the planning and execution of pivotal studies addressing COVID-19. Our objective was to summarize the results of these collaborations and lessons learned.
Results:The TIN provided 29 COVID-related consults between March 2020 and December 2020, including 6 trial participation expressions of interest and 8 community engagement studios from the Recruitment Innovation Center. Key lessons learned from these experiences include the benefits of leveraging an established infrastructure, innovations surrounding remote research activities, data harmonization and central safety reviews, and early community engagement and involvement.
Conclusions:Our experience highlighted the benefits and challenges of a multi-institutional approach to clinical research during a pandemic.
Reanalysis of the Atmospheric Radiocarbon Calibration Record from Lake Suigetsu, Japan
- Part of
- Christopher Bronk Ramsey, Timothy J Heaton, Gordon Schlolaut, Richard A Staff, Charlotte L Bryant, Achim Brauer, Henry F Lamb, Michael H Marshall, Takeshi Nakagawa
-
- Journal:
- Radiocarbon / Volume 62 / Issue 4 / August 2020
- Published online by Cambridge University Press:
- 25 March 2020, pp. 989-999
- Print publication:
- August 2020
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Terrestrial plant macrofossils from the sedimentary record of Lake Suigetsu, Japan, provide the only quasi-continuous direct atmospheric record of radiocarbon (14C) covering the last 50 ka cal BP (Bronk Ramsey et al. 2012). Since then, new high precision data have become available on U-Th dated speleothems from Hulu Cave China, covering the same time range (Cheng et al. 2018). In addition, an updated varve-based chronology has also been published for the 2006 core from Lake Suigetsu (SG06) based on extended microscopic analysis of the sediments and improved algorithms for interpolation (Schlolaut et al. 2018). Here we reanalyze the radiocarbon dataset from Suigetsu based on the new varve counting information and the constraints imposed by the speleothem data. This enables the new information on the calendar age scale of the Suigetsu dataset to be used in the construction of the consensus IntCal calibration curve. Comparison of the speleothem and plant macrofossil records provides insight into the mechanisms underlying the incorporation of carbon into different types of record and the relative strengths of different types of archive for calibration purposes.
A collaborative, academic approach to optimizing the national clinical research infrastructure: The first year of the Trial Innovation Network
- Gordon R. Bernard, Paul A. Harris, Jill M. Pulley, Daniel K. Benjamin, Jonathan Michael Dean, Daniel E. Ford, Daniel F. Hanley, Harry P. Selker, Consuelo H. Wilkins
-
- Journal:
- Journal of Clinical and Translational Science / Volume 2 / Issue 4 / August 2018
- Published online by Cambridge University Press:
- 27 November 2018, pp. 187-192
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
-
Inefficiencies in the national clinical research infrastructure have been apparent for decades. The National Center for Advancing Translational Science—sponsored Clinical and Translational Science Award (CTSA) program is able to address such inefficiencies. The Trial Innovation Network (TIN) is a collaborative initiative with the CTSA program and other National Institutes of Health (NIH) Institutes and Centers that addresses critical roadblocks to accelerate the translation of novel interventions to clinical practice. The TIN’s mission is to execute high-quality trials in a quick, cost-efficient manner. The TIN awardees are composed of 3 Trial Innovation Centers, the Recruitment Innovation Center, and the individual CTSA institutions that have identified TIN Liaison units. The TIN has launched a national scale single (central) Institutional Review Board system, master contracting agreements, quality-by-design approaches, novel recruitment support methods, and applies evidence-based strategies to recruitment and patient engagement. The TIN has received 113 submissions from 39 different CTSA institutions and 8 non-CTSA Institutions, with projects associated with 12 different NIH Institutes and Centers across a wide range of clinical/disease areas. Already more than 150 unique health systems/organizations are involved as sites in TIN-related multisite studies. The TIN will begin to capture data and metrics that quantify increased efficiency and quality improvement during operations.
The Taipan Galaxy Survey: Scientific Goals and Observing Strategy
- Elisabete da Cunha, Andrew M. Hopkins, Matthew Colless, Edward N. Taylor, Chris Blake, Cullan Howlett, Christina Magoulas, John R. Lucey, Claudia Lagos, Kyler Kuehn, Yjan Gordon, Dilyar Barat, Fuyan Bian, Christian Wolf, Michael J. Cowley, Marc White, Ixandra Achitouv, Maciej Bilicki, Joss Bland-Hawthorn, Krzysztof Bolejko, Michael J. I. Brown, Rebecca Brown, Julia Bryant, Scott Croom, Tamara M. Davis, Simon P. Driver, Miroslav D. Filipovic, Samuel R. Hinton, Melanie Johnston-Hollitt, D. Heath Jones, Bärbel Koribalski, Dane Kleiner, Jon Lawrence, Nuria Lorente, Jeremy Mould, Matt S. Owers, Kevin Pimbblet, C. G. Tinney, Nicholas F. H. Tothill, Fred Watson
-
- Journal:
- Publications of the Astronomical Society of Australia / Volume 34 / 2017
- Published online by Cambridge University Press:
- 24 October 2017, e047
-
- Article
-
- You have access Access
- HTML
- Export citation
-
The Taipan galaxy survey (hereafter simply ‘Taipan’) is a multi-object spectroscopic survey starting in 2017 that will cover 2π steradians over the southern sky (δ ≲ 10°, |b| ≳ 10°), and obtain optical spectra for about two million galaxies out to z < 0.4. Taipan will use the newly refurbished 1.2-m UK Schmidt Telescope at Siding Spring Observatory with the new TAIPAN instrument, which includes an innovative ‘Starbugs’ positioning system capable of rapidly and simultaneously deploying up to 150 spectroscopic fibres (and up to 300 with a proposed upgrade) over the 6° diameter focal plane, and a purpose-built spectrograph operating in the range from 370 to 870 nm with resolving power R ≳ 2000. The main scientific goals of Taipan are (i) to measure the distance scale of the Universe (primarily governed by the local expansion rate, H0) to 1% precision, and the growth rate of structure to 5%; (ii) to make the most extensive map yet constructed of the total mass distribution and motions in the local Universe, using peculiar velocities based on improved Fundamental Plane distances, which will enable sensitive tests of gravitational physics; and (iii) to deliver a legacy sample of low-redshift galaxies as a unique laboratory for studying galaxy evolution as a function of dark matter halo and stellar mass and environment. The final survey, which will be completed within 5 yrs, will consist of a complete magnitude-limited sample (i ⩽ 17) of about 1.2 × 106 galaxies supplemented by an extension to higher redshifts and fainter magnitudes (i ⩽ 18.1) of a luminous red galaxy sample of about 0.8 × 106 galaxies. Observations and data processing will be carried out remotely and in a fully automated way, using a purpose-built automated ‘virtual observer’ software and an automated data reduction pipeline. The Taipan survey is deliberately designed to maximise its legacy value by complementing and enhancing current and planned surveys of the southern sky at wavelengths from the optical to the radio; it will become the primary redshift and optical spectroscopic reference catalogue for the local extragalactic Universe in the southern sky for the coming decade.
Contributors
-
- By Mitchell Aboulafia, Frederick Adams, Marilyn McCord Adams, Robert M. Adams, Laird Addis, James W. Allard, David Allison, William P. Alston, Karl Ameriks, C. Anthony Anderson, David Leech Anderson, Lanier Anderson, Roger Ariew, David Armstrong, Denis G. Arnold, E. J. Ashworth, Margaret Atherton, Robin Attfield, Bruce Aune, Edward Wilson Averill, Jody Azzouni, Kent Bach, Andrew Bailey, Lynne Rudder Baker, Thomas R. Baldwin, Jon Barwise, George Bealer, William Bechtel, Lawrence C. Becker, Mark A. Bedau, Ernst Behler, José A. Benardete, Ermanno Bencivenga, Jan Berg, Michael Bergmann, Robert L. Bernasconi, Sven Bernecker, Bernard Berofsky, Rod Bertolet, Charles J. Beyer, Christian Beyer, Joseph Bien, Joseph Bien, Peg Birmingham, Ivan Boh, James Bohman, Daniel Bonevac, Laurence BonJour, William J. Bouwsma, Raymond D. Bradley, Myles Brand, Richard B. Brandt, Michael E. Bratman, Stephen E. Braude, Daniel Breazeale, Angela Breitenbach, Jason Bridges, David O. Brink, Gordon G. Brittan, Justin Broackes, Dan W. Brock, Aaron Bronfman, Jeffrey E. Brower, Bartosz Brozek, Anthony Brueckner, Jeffrey Bub, Lara Buchak, Otavio Bueno, Ann E. Bumpus, Robert W. Burch, John Burgess, Arthur W. Burks, Panayot Butchvarov, Robert E. Butts, Marina Bykova, Patrick Byrne, David Carr, Noël Carroll, Edward S. Casey, Victor Caston, Victor Caston, Albert Casullo, Robert L. Causey, Alan K. L. Chan, Ruth Chang, Deen K. Chatterjee, Andrew Chignell, Roderick M. Chisholm, Kelly J. Clark, E. J. Coffman, Robin Collins, Brian P. Copenhaver, John Corcoran, John Cottingham, Roger Crisp, Frederick J. Crosson, Antonio S. Cua, Phillip D. Cummins, Martin Curd, Adam Cureton, Andrew Cutrofello, Stephen Darwall, Paul Sheldon Davies, Wayne A. Davis, Timothy Joseph Day, Claudio de Almeida, Mario De Caro, Mario De Caro, John Deigh, C. F. Delaney, Daniel C. Dennett, Michael R. DePaul, Michael Detlefsen, Daniel Trent Devereux, Philip E. Devine, John M. Dillon, Martin C. Dillon, Robert DiSalle, Mary Domski, Alan Donagan, Paul Draper, Fred Dretske, Mircea Dumitru, Wilhelm Dupré, Gerald Dworkin, John Earman, Ellery Eells, Catherine Z. Elgin, Berent Enç, Ronald P. Endicott, Edward Erwin, John Etchemendy, C. Stephen Evans, Susan L. Feagin, Solomon Feferman, Richard Feldman, Arthur Fine, Maurice A. Finocchiaro, William FitzPatrick, Richard E. Flathman, Gvozden Flego, Richard Foley, Graeme Forbes, Rainer Forst, Malcolm R. Forster, Daniel Fouke, Patrick Francken, Samuel Freeman, Elizabeth Fricker, Miranda Fricker, Michael Friedman, Michael Fuerstein, Richard A. Fumerton, Alan Gabbey, Pieranna Garavaso, Daniel Garber, Jorge L. A. Garcia, Robert K. Garcia, Don Garrett, Philip Gasper, Gerald Gaus, Berys Gaut, Bernard Gert, Roger F. Gibson, Cody Gilmore, Carl Ginet, Alan H. Goldman, Alvin I. Goldman, Alfonso Gömez-Lobo, Lenn E. Goodman, Robert M. Gordon, Stefan Gosepath, Jorge J. E. Gracia, Daniel W. Graham, George A. Graham, Peter J. Graham, Richard E. Grandy, I. Grattan-Guinness, John Greco, Philip T. Grier, Nicholas Griffin, Nicholas Griffin, David A. Griffiths, Paul J. Griffiths, Stephen R. Grimm, Charles L. Griswold, Charles B. Guignon, Pete A. Y. Gunter, Dimitri Gutas, Gary Gutting, Paul Guyer, Kwame Gyekye, Oscar A. Haac, Raul Hakli, Raul Hakli, Michael Hallett, Edward C. Halper, Jean Hampton, R. James Hankinson, K. R. Hanley, Russell Hardin, Robert M. Harnish, William Harper, David Harrah, Kevin Hart, Ali Hasan, William Hasker, John Haugeland, Roger Hausheer, William Heald, Peter Heath, Richard Heck, John F. Heil, Vincent F. Hendricks, Stephen Hetherington, Francis Heylighen, Kathleen Marie Higgins, Risto Hilpinen, Harold T. Hodes, Joshua Hoffman, Alan Holland, Robert L. Holmes, Richard Holton, Brad W. Hooker, Terence E. Horgan, Tamara Horowitz, Paul Horwich, Vittorio Hösle, Paul Hoβfeld, Daniel Howard-Snyder, Frances Howard-Snyder, Anne Hudson, Deal W. Hudson, Carl A. Huffman, David L. Hull, Patricia Huntington, Thomas Hurka, Paul Hurley, Rosalind Hursthouse, Guillermo Hurtado, Ronald E. Hustwit, Sarah Hutton, Jonathan Jenkins Ichikawa, Harry A. Ide, David Ingram, Philip J. Ivanhoe, Alfred L. Ivry, Frank Jackson, Dale Jacquette, Joseph Jedwab, Richard Jeffrey, David Alan Johnson, Edward Johnson, Mark D. Jordan, Richard Joyce, Hwa Yol Jung, Robert Hillary Kane, Tomis Kapitan, Jacquelyn Ann K. Kegley, James A. Keller, Ralph Kennedy, Sergei Khoruzhii, Jaegwon Kim, Yersu Kim, Nathan L. King, Patricia Kitcher, Peter D. Klein, E. D. Klemke, Virginia Klenk, George L. Kline, Christian Klotz, Simo Knuuttila, Joseph J. Kockelmans, Konstantin Kolenda, Sebastian Tomasz Kołodziejczyk, Isaac Kramnick, Richard Kraut, Fred Kroon, Manfred Kuehn, Steven T. Kuhn, Henry E. Kyburg, John Lachs, Jennifer Lackey, Stephen E. Lahey, Andrea Lavazza, Thomas H. Leahey, Joo Heung Lee, Keith Lehrer, Dorothy Leland, Noah M. Lemos, Ernest LePore, Sarah-Jane Leslie, Isaac Levi, Andrew Levine, Alan E. Lewis, Daniel E. Little, Shu-hsien Liu, Shu-hsien Liu, Alan K. L. Chan, Brian Loar, Lawrence B. Lombard, John Longeway, Dominic McIver Lopes, Michael J. Loux, E. J. Lowe, Steven Luper, Eugene C. Luschei, William G. Lycan, David Lyons, David Macarthur, Danielle Macbeth, Scott MacDonald, Jacob L. Mackey, Louis H. Mackey, Penelope Mackie, Edward H. Madden, Penelope Maddy, G. B. Madison, Bernd Magnus, Pekka Mäkelä, Rudolf A. Makkreel, David Manley, William E. Mann (W.E.M.), Vladimir Marchenkov, Peter Markie, Jean-Pierre Marquis, Ausonio Marras, Mike W. Martin, A. P. Martinich, William L. McBride, David McCabe, Storrs McCall, Hugh J. McCann, Robert N. McCauley, John J. McDermott, Sarah McGrath, Ralph McInerny, Daniel J. McKaughan, Thomas McKay, Michael McKinsey, Brian P. McLaughlin, Ernan McMullin, Anthonie Meijers, Jack W. Meiland, William Jason Melanson, Alfred R. Mele, Joseph R. Mendola, Christopher Menzel, Michael J. Meyer, Christian B. Miller, David W. Miller, Peter Millican, Robert N. Minor, Phillip Mitsis, James A. Montmarquet, Michael S. Moore, Tim Moore, Benjamin Morison, Donald R. Morrison, Stephen J. Morse, Paul K. Moser, Alexander P. D. Mourelatos, Ian Mueller, James Bernard Murphy, Mark C. Murphy, Steven Nadler, Jan Narveson, Alan Nelson, Jerome Neu, Samuel Newlands, Kai Nielsen, Ilkka Niiniluoto, Carlos G. Noreña, Calvin G. Normore, David Fate Norton, Nikolaj Nottelmann, Donald Nute, David S. Oderberg, Steve Odin, Michael O’Rourke, Willard G. Oxtoby, Heinz Paetzold, George S. Pappas, Anthony J. Parel, Lydia Patton, R. P. Peerenboom, Francis Jeffry Pelletier, Adriaan T. Peperzak, Derk Pereboom, Jaroslav Peregrin, Glen Pettigrove, Philip Pettit, Edmund L. Pincoffs, Andrew Pinsent, Robert B. Pippin, Alvin Plantinga, Louis P. Pojman, Richard H. Popkin, John F. Post, Carl J. Posy, William J. Prior, Richard Purtill, Michael Quante, Philip L. Quinn, Philip L. Quinn, Elizabeth S. Radcliffe, Diana Raffman, Gerard Raulet, Stephen L. Read, Andrews Reath, Andrew Reisner, Nicholas Rescher, Henry S. Richardson, Robert C. Richardson, Thomas Ricketts, Wayne D. Riggs, Mark Roberts, Robert C. Roberts, Luke Robinson, Alexander Rosenberg, Gary Rosenkranz, Bernice Glatzer Rosenthal, Adina L. Roskies, William L. Rowe, T. M. Rudavsky, Michael Ruse, Bruce Russell, Lilly-Marlene Russow, Dan Ryder, R. M. Sainsbury, Joseph Salerno, Nathan Salmon, Wesley C. Salmon, Constantine Sandis, David H. Sanford, Marco Santambrogio, David Sapire, Ruth A. Saunders, Geoffrey Sayre-McCord, Charles Sayward, James P. Scanlan, Richard Schacht, Tamar Schapiro, Frederick F. Schmitt, Jerome B. Schneewind, Calvin O. Schrag, Alan D. Schrift, George F. Schumm, Jean-Loup Seban, David N. Sedley, Kenneth Seeskin, Krister Segerberg, Charlene Haddock Seigfried, Dennis M. Senchuk, James F. Sennett, William Lad Sessions, Stewart Shapiro, Tommie Shelby, Donald W. Sherburne, Christopher Shields, Roger A. Shiner, Sydney Shoemaker, Robert K. Shope, Kwong-loi Shun, Wilfried Sieg, A. John Simmons, Robert L. Simon, Marcus G. Singer, Georgette Sinkler, Walter Sinnott-Armstrong, Matti T. Sintonen, Lawrence Sklar, Brian Skyrms, Robert C. Sleigh, Michael Anthony Slote, Hans Sluga, Barry Smith, Michael Smith, Robin Smith, Robert Sokolowski, Robert C. Solomon, Marta Soniewicka, Philip Soper, Ernest Sosa, Nicholas Southwood, Paul Vincent Spade, T. L. S. Sprigge, Eric O. Springsted, George J. Stack, Rebecca Stangl, Jason Stanley, Florian Steinberger, Sören Stenlund, Christopher Stephens, James P. Sterba, Josef Stern, Matthias Steup, M. A. Stewart, Leopold Stubenberg, Edith Dudley Sulla, Frederick Suppe, Jere Paul Surber, David George Sussman, Sigrún Svavarsdóttir, Zeno G. Swijtink, Richard Swinburne, Charles C. Taliaferro, Robert B. Talisse, John Tasioulas, Paul Teller, Larry S. Temkin, Mark Textor, H. S. Thayer, Peter Thielke, Alan Thomas, Amie L. Thomasson, Katherine Thomson-Jones, Joshua C. Thurow, Vzalerie Tiberius, Terrence N. Tice, Paul Tidman, Mark C. Timmons, William Tolhurst, James E. Tomberlin, Rosemarie Tong, Lawrence Torcello, Kelly Trogdon, J. D. Trout, Robert E. Tully, Raimo Tuomela, John Turri, Martin M. Tweedale, Thomas Uebel, Jennifer Uleman, James Van Cleve, Harry van der Linden, Peter van Inwagen, Bryan W. Van Norden, René van Woudenberg, Donald Phillip Verene, Samantha Vice, Thomas Vinci, Donald Wayne Viney, Barbara Von Eckardt, Peter B. M. Vranas, Steven J. Wagner, William J. Wainwright, Paul E. Walker, Robert E. Wall, Craig Walton, Douglas Walton, Eric Watkins, Richard A. Watson, Michael V. Wedin, Rudolph H. Weingartner, Paul Weirich, Paul J. Weithman, Carl Wellman, Howard Wettstein, Samuel C. Wheeler, Stephen A. White, Jennifer Whiting, Edward R. Wierenga, Michael Williams, Fred Wilson, W. Kent Wilson, Kenneth P. Winkler, John F. Wippel, Jan Woleński, Allan B. Wolter, Nicholas P. Wolterstorff, Rega Wood, W. Jay Wood, Paul Woodruff, Alison Wylie, Gideon Yaffe, Takashi Yagisawa, Yutaka Yamamoto, Keith E. Yandell, Xiaomei Yang, Dean Zimmerman, Günter Zoller, Catherine Zuckert, Michael Zuckert, Jack A. Zupko (J.A.Z.)
- Edited by Robert Audi, University of Notre Dame, Indiana
-
- Book:
- The Cambridge Dictionary of Philosophy
- Published online:
- 05 August 2015
- Print publication:
- 27 April 2015, pp ix-xxx
-
- Chapter
- Export citation
Contributors
-
- By Rony A. Adam, Gloria Bachmann, Nichole M. Barker, Randall B. Barnes, John Bennett, Inbar Ben-Shachar, Jonathan S. Berek, Sarah L. Berga, Monica W. Best, Eric J. Bieber, Frank M. Biro, Shan Biscette, Anita K. Blanchard, Candace Brown, Ronald T. Burkman, Joseph Buscema, John E. Buster, Michael Byas-Smith, Sandra Ann Carson, Judy C. Chang, Annie N. Y. Cheung, Mindy S. Christianson, Karishma Circelli, Daniel L. Clarke-Pearson, Larry J. Copeland, Bryan D. Cowan, Navneet Dhillon, Michael P. Diamond, Conception Diaz-Arrastia, Nicole M. Donnellan, Michael L. Eisenberg, Eric Eisenhauer, Sebastian Faro, J. Stuart Ferriss, Lisa C. Flowers, Susan J. Freeman, Leda Gattoc, Claudine Marie Gayle, Timothy M. Geiger, Jennifer S. Gell, Alan N. Gordon, Victoria L. Green, Jon K. Hathaway, Enrique Hernandez, S. Paige Hertweck, Randall S. Hines, Ira R. Horowitz, Fred M. Howard, William W. Hurd, Fidan Israfilbayli, Denise J. Jamieson, Carolyn R. Jaslow, Erika B. Johnston-MacAnanny, Rohna M. Kearney, Namita Khanna, Caroline C. King, Jeremy A. King, Ira J. Kodner, Tamara Kolev, Athena P. Kourtis, S. Robert Kovac, Ertug Kovanci, William H. Kutteh, Eduardo Lara-Torre, Pallavi Latthe, Herschel W. Lawson, Ronald L. Levine, Frank W. Ling, Larry I. Lipshultz, Steven D. McCarus, Robert McLellan, Shruti Malik, Suketu M. Mansuria, Mohamed K. Mehasseb, Pamela J. Murray, Saloney Nazeer, Farr R. Nezhat, Hextan Y. S. Ngan, Gina M. Northington, Peggy A. Norton, Ruth M. O'Regan, Kristiina Parviainen, Resad P. Pasic, Tanja Pejovic, K. Ulrich Petry, Nancy A. Phillips, Ashish Pradhan, Elizabeth E. Puscheck, Suneetha Rachaneni, Devon M. Ramaeker, David B. Redwine, Robert L. Reid, Carla P. Roberts, Walter Romano, Peter G. Rose, Robert L. Rosenfield, Shon P. Rowan, Mack T. Ruffin, Janice M. Rymer, Evis Sala, Ritu Salani, Joseph S. Sanfilippo, Mahmood I. Shafi, Roger P. Smith, Meredith L. Snook, Thomas E. Snyder, Mary D. Stephenson, Thomas G. Stovall, Richard L. Sweet, Philip M. Toozs-Hobson, Togas Tulandi, Elizabeth R. Unger, Denise S. Uyar, Marion S. Verp, Rahi Victory, Tamara J. Vokes, Michelle J. Washington, Katharine O'Connell White, Paul E. Wise, Frank M. Wittmaack, Miya P. Yamamoto, Christine Yu, Howard A. Zacur
- Edited by Eric J. Bieber, Joseph S. Sanfilippo, University of Pittsburgh, Ira R. Horowitz, Emory University, Atlanta, Mahmood I. Shafi
-
- Book:
- Clinical Gynecology
- Published online:
- 05 April 2015
- Print publication:
- 23 April 2015, pp viii-xiv
-
- Chapter
- Export citation
Good is not Good Enough: The Benchmark Stroke Door-to-Needle Time Should be 30 Minutes
- Noreen Kamal, Oscar Benavente, Karl Boyle, Brian Buck, Ken Butcher, Leanne K. Casaubon, Robert Côté, Andrew M Demchuk, Yan Deschaintre, Dar Dowlatshahi, Gordon J Gubitz, Gary Hunter, Tom Jeerakathil, Albert Jin, Eddy Lang, Sylvain Lanthier, Patrice Lindsay, Nancy Newcommon, Jennifer Mandzia, Colleen M. Norris, Wes Oczkowski, Céline Odier, Stephen Phillips, Alexandre Y Poppe, Gustavo Saposnik, Daniel Selchen, Ashfaq Shuaib, Frank Silver, Eric E Smith, Grant Stotts, Michael Suddes, Richard H. Swartz, Philip Teal, Tim Watson, Michael D. Hill
-
- Journal:
- Canadian Journal of Neurological Sciences / Volume 41 / Issue 6 / November 2014
- Published online by Cambridge University Press:
- 20 October 2014, pp. 694-696
-
- Article
-
- You have access Access
- HTML
- Export citation
Contributors
-
- By Shyamkrishna Balganesh, Dan L. Burk, Eric R. Claeys, Thomas F. Cotter, Hanoch Dagan, Richard A. Epstein, Jeanne C. Fromer, Wendy J. Gordon, Paul J. Heald, Steven Hetcher, David Lametti, Mark A. Lemley, Margaret H. Lemos, Mark P. McKenna, Peter S. Menell, Gideon Parchomovsky, Lee Petherbridge, Michael Risch, Jennifer E. Rothman, Emily Sherwin, Henry E. Smith, Madhavi Sunder, Molly Shaffer Van Houweling, R. Polk Wagner, Christopher S. Yoo
- Edited by Shyamkrishna Balganesh, University of Pennsylvania Law School
-
- Book:
- Intellectual Property and the Common Law
- Published online:
- 05 September 2013
- Print publication:
- 02 September 2013, pp ix-x
-
- Chapter
- Export citation
Contributors
-
- By Albert Altchek, David H. Barad, Katharine Batt, Yuval Bdolah, Revaz Botchorishvili, Nicolas Bourdel, Michael S. Broder, Douglas N. Brown, Jubilee Brown, Antoine Maurice Bruhat, Michel Canis, Mine S. Cicek, Carmel J. Cohen, Christopher P. Crum, Christina E. Curtin, Liane Deligdisch, Philip J. Di Saia, Ramez N. Eskander, Tamara Finger, David Fishman, Brooke L. Fridley, David M. Gershenson, Norbert Gleicher, Ellen L. Goode, Pierre S. Gordon, Ioannis Gryparis, Jonathan Hecht, Wendy C. Hsiao, Eric C. Huang, Nathan G. Kase, Valentin Kolev, Lale Kostakoglu, Neri Laufer, Anna Laury, Gerard Mage, Angelica Mareş, Maurie Markman, Luciano G. Nardo, Farr R. Nezhat, Sree Durga Patchava, Tanja Pejovic, Catherine M. Phelan, Benoit Rabischong, Jamal Rahaman, David Rodriguez-Buritica, Paul Saenger, Peter Schlosshauer, William L. Simpson, Cardinale B. Smith, Jason Sternchos
- Edited by Liane Deligdisch, Nathan G. Kase, Carmel J. Cohen
-
- Book:
- Altchek's Diagnosis and Management of Ovarian Disorders
- Published online:
- 05 August 2013
- Print publication:
- 25 July 2013, pp vii-x
-
- Chapter
- Export citation
Contributors
-
- By Victoria M. Allen, Frederic Amant, Sarah Armstrong, Thomas F. Baskett, Michael A. Belfort, Meredith Birsner, Renee D. Boss, Leanne Bricker, Josaphat K. Byamugisha, Giorgio Capogna, Michael P. Casaer, Frank A. Chervenak, Vicki Clark, Filip Claus, Malachy O. Columb, Charles Cox, Jean T. Cox, Vegard Dahl, John Davison, Jan Deprest, Clifford S. Deutschman, Roland Devlieger, Karim Djekidel, Steven Dymarkowski, Roshan Fernando, Clare Fitzpatrick, Sreedhar Gaddipati, Thierry Girard, Emily Gordon, Ian A. Greer, David Grooms, Sina Haeri, Katy Harrison, Edward J. Hayes, Michelle Hladunewich, Andra H. James, Tracey Johnston, Bellal Joseph, Erin Keely, Ruth Landau, Stephen E. Lapinsky, Susanna I. Lee, Larry Leeman, Hennie Lombaard, Stephen Lu, Alison MacArthur, Laura A. Magee, Paul E. Marik, Laurence B. McCullough, Alexandre Mignon, Carlo Missant, Jack Moodley, Lisa E. Moore, Kate Morse, Warwick D. Ngan Kee, Catherine Nelson-Piercy, Clemens M. Ortner, Geraldine O’Sullivan, Luis D. Pacheco, Fathima Paruk, Melina Pectasides, Nigel Pereira, Patricia Peticca, Sharon T. Phelan, Felicity Plaat, Lauren A. Plante, Michael P. Plevyak, Dianne Plews, Wendy Pollock, Laura C. Price, Peter Rhee, Leiv Arne Rosseland, Kathryn M. Rowan, Helen Ryan, Helen Scholefield, Neil S. Seligman, Nadir Sharawi, Alex Sia, Bob Silver, Mieke Soens, Ulrich J. Spreng, Silvia Stirparo, Nova Szoka, Andrew Tang, Kha M. Tran, Els Troost, Lawrence C. Tsen, Derek Tuffnell, Kristel Van Calsteren, Marc Van de Velde, Marcel Vercauteren, Chris Verslype, Peter von Dadelszen, Carl Waldman, Michelle Walters, Linda Watkins, Paul Westhead, Cynthia A. Wong, Gerda G. Zeeman, Joost J. Zwart
- Edited by Marc van de Velde, Helen Scholefield, Lauren A. Plante
-
- Book:
- Maternal Critical Care
- Published online:
- 05 July 2013
- Print publication:
- 04 July 2013, pp ix-xiv
-
- Chapter
- Export citation
Contributors
-
- By Janine B. Adams, Kirsten B. Barnes, Guy C. Bate, Greg A. Botha, Meyrick B. Bowker, Sarah J. Bownes, Nicola K. Carrasco, Clinton P. Chrystal, Robynne A. Chrystal, Xander Combrink, Allan D. Connell, Digby P. Cyrus, Colleen T. Downs, William N. Ellery, Anthony T. Forbes, Nicolette T. Forbes, Caroline Fox, Nuette Gordon, Michael C. Grenfell, Suzanne E. Grenfell, Sylvi Haldorsen, Marc S. Humphries, Hendrik L. Jerling, Bruce E. Kelbe, C. Fiona MacKay, Christopher M. Maine, Andrew Z. Maro, Andrew A. Mather, Nelson A. F. Miranda, David G. Muir, Holly A. Nel, Sibulele Nondoda, Renzo Perissinotto, Deena Pillay, Naomi Porat, Roger N. Porter, Sean N. Porter, Justin J. Pringle, Ursula M. Scharler, Derek D. Stretch, Ricky H. Taylor, Jane Turpie, Jonathan K. Warner, Alan K. Whitfield
- Edited by Renzo Perissinotto, University of KwaZulu-Natal, South Africa, Derek D. Stretch, University of KwaZulu-Natal, South Africa, Ricky H. Taylor
-
- Book:
- Ecology and Conservation of Estuarine Ecosystems
- Published online:
- 05 April 2013
- Print publication:
- 16 May 2013, pp xiii-xvi
-
- Chapter
- Export citation
Integration of the Old and New Lake Suigetsu (Japan) Terrestrial Radiocarbon Calibration Data Sets
- Richard A Staff, Gordon Schlolaut, Christopher Bronk Ramsey, Fiona Brock, Charlotte L Bryant, Hiroyuki Kitagawa, Johannes van der Plicht, Michael H Marshall, Achim Brauer, Henry F Lamb, Rebecca L Payne, Pavel E Tarasov, Tsuyoshi Haraguchi, Katsuya Gotanda, Hitoshi Yonenobu, Yusuke Yokoyama, Takeshi Nakagawa, Suigetsu 2006 Project Members
-
- Journal:
- Radiocarbon / Volume 55 / Issue 4 / 2013
- Published online by Cambridge University Press:
- 09 February 2016, pp. 2049-2058
- Print publication:
- 2013
-
- Article
-
- You have access Access
- Export citation
-
The varved sediment profile of Lake Suigetsu, central Japan, offers an ideal opportunity from which to derive a terrestrial record of atmospheric radiocarbon across the entire range of the 14C dating method. Previous work by Kitagawa and van der Plicht (1998a,b, 2000) provided such a data set; however, problems with the varve-based age scale of their SG93 sediment core precluded the use of this data set for 14C calibration purposes. Lake Suigetsu was re-cored in summer 2006, with the retrieval of overlapping sediment cores from 4 parallel boreholes enabling complete recovery of the sediment profile for the present “Suigetsu Varves 2006” project (Nakagawa et al. 2012). Over 550 14C determinations have been obtained from terrestrial plant macrofossils picked from the latter SG06 composite sediment core, which, coupled with the core's independent varve chronology, provides the only non-reservoir-corrected 14C calibration data set across the 14C dating range.
Here, physical matching of archive U-channel sediment from SG93 to the continuous SG06 sediment profile is presented. We show the excellent agreement between the respective projects' 14C data sets, allowing the integration of 243 14C determinations from the original SG93 project into a composite Lake Suigetsu 14C calibration data set comprising 808 individual 14C determinations, spanning the last 52,800 cal yr.
Effects of acute consumption of a fruit and vegetable purée-based drink on vasodilation and oxidative status
- Trevor W. George, Saran Waroonphan, Chutamat Niwat, Michael H. Gordon, Julie A. Lovegrove
-
- Journal:
- British Journal of Nutrition / Volume 109 / Issue 8 / 28 April 2013
- Published online by Cambridge University Press:
- 28 September 2012, pp. 1442-1452
- Print publication:
- 28 April 2013
-
- Article
-
- You have access Access
- HTML
- Export citation
-
Epidemiological studies indicate that diets rich in fruits and vegetables (F&V) are protective against CVD. Puréed F&V products retain many beneficial components, including flavonoids, carotenoids, vitamin C and dietary fibres. The present study aimed to establish the physiological effects of acute ingestion of a F&V purée-based drink (FVPD) on vasodilation, antioxidant status, phytochemical bioavailability and other CVD risk factors. A total of twenty-four subjects, aged 30–70 years, completed the randomised, single-blind, controlled, crossover test meal study. Subjects consumed 400 ml of the FVPD, or a fruit-flavoured sugar-matched control, after following a low-flavonoid diet for 5 d. Blood and urine samples were collected throughout the study day, and vascular reactivity was assessed at 90 min intervals using laser Doppler iontophoresis. The FVPD significantly increased plasma vitamin C (P= 0·002) and total nitrate/nitrite (P= 0·001) concentrations. There was a near significant time by treatment effect on ex vivo LDL oxidation (P= 0·068), with a longer lag phase after consuming the FVPD. During the 6 h after juice consumption, the antioxidant capacity of plasma increased significantly (P= 0·003) and there was a simultaneous increase in plasma and urinary phenolic metabolites (P< 0·05). There were significantly lower glucose and insulin peaks after ingestion of the FVPD compared with control (P= 0·019 and 0·003) and a trend towards increased endothelium-dependent vasodilation following FVPD consumption (P= 0·061). Overall, FVPD consumption significantly increased plasma vitamin C and total nitrate/nitrite concentrations, with a trend towards increased endothelium-dependent vasodilation. Puréed F&V products are useful vehicles for increasing micronutrient status, plasma antioxidant capacity and in vivo NO generation, which may contribute to CVD risk reduction.